Safety, Tolerability, and Efficacy of Immunomodulation w/a Monoclonal Antibody Against CD40L in Combination With Transplanted Islet Cells in Adults With"Brittle" T1D-Piotr Witkowski MD PhD
Ahead of the March 31 talk
T1D Research Talk Series · TheSugarScience
Piotr Witkowski MD PhD presents -A clinical trial update from the University of Chicago — and why this could change islet transplantation forever
📅 Registration
Tuesday, March 31, 2026 · 12:00 PM ET
9:00 AM Pacific · Free & open to all
👤 Speaker
Piotr Witkowski, MD, PhD
Director, Pancreas and Islet Transplant Program
University of Chicago Medicine’s Transplant Institute
Dr. Witkowski is one of the world’s leading experts in islet transplantation, with additional expertise in kidney, pancreatic, and liver transplantation. He was instrumental in developing an optimized islet isolation technique that greatly improved clinical transplant success. Under his leadership at the University of Chicago, multidisciplinary teams are conducting studies designed to improve quality and outcomes in islet cell transplantation in patients with T1D — including the first clinical trial using tegoprubart as a calcineurin inhibitor-free immunosuppressive regimen following islet transplant.
01 · The Clinical Trial
Summary of the Science
“Safety, Tolerability, and Efficacy of Immunomodulation With a Monoclonal Antibody Against CD40L in Combination With Transplanted Islet Cells in Adults With Brittle Type 1 Diabetes Mellitus”
Witkowski P et al. · University of Chicago Medicine · NCT05480657 · Phase 1/2
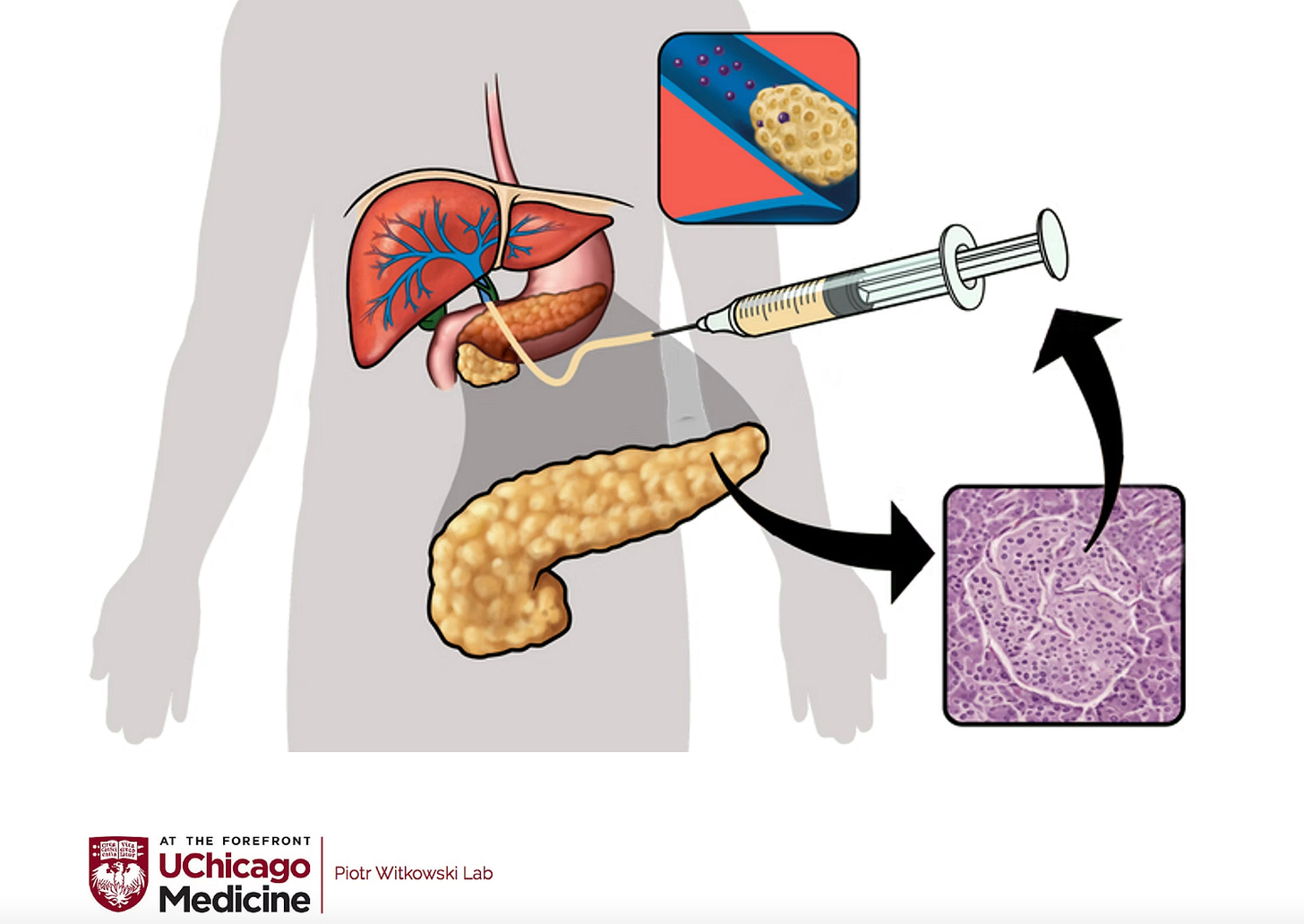
For over 30 years, islet transplantation has offered a path to insulin independence for patients with the most severe, unstable forms of T1D — but it has been held back by one stubborn problem: the toxicity of the immunosuppressive drugs needed to prevent graft rejection. The current standard of care, tacrolimus, is effective but damages the kidneys, the nervous system, the cardiovascular system, and — critically — the islet cells themselves.
This trial asks a direct and urgent question: can tegoprubart, a second-generation anti-CD40L monoclonal antibody developed by Eledon Pharmaceuticals, replace tacrolimus as the core immunosuppressant following islet transplantation — offering equivalent or better graft protection with a dramatically safer profile?
Tegoprubart works by blocking CD40 ligand (CD40L/CD154), a protein that acts as a master switch for immune cell activation. By interrupting the CD40–CD40L costimulatory pathway, tegoprubart simultaneously inhibits T cell activation, suppresses B cell responses, and — crucially — increases the polarization of lymphocytes into regulatory T cells (Tregs) that actively promote immune tolerance. Unlike tacrolimus, which broadly suppresses immune function and is directly toxic to beta cells, tegoprubart takes a targeted, non-lymphocyte-depleting approach.
The trial enrolled adults with “brittle” T1D — patients with severely unstable blood glucose, frequent dangerous hypoglycemic events, and hypoglycemia unawareness despite optimal insulin management. These are patients for whom standard diabetes management has failed and whose quality of life is severely compromised. Participants received deceased-donor islet cell infusions into the liver, followed by intravenous tegoprubart infusions every three weeks as their core immunosuppression.
The results have been remarkable. All six subjects in the initial cohort achieved insulin independence after one or two transplants, with sustained normal HbA1c. Islet engraftment in the first two tegoprubart-treated subjects was estimated to be three to five times higher than in comparable subjects receiving tacrolimus — strongly suggesting that removing tacrolimus toxicity allows the transplanted islets to survive and function far better. No subjects experienced severe hypoglycemia, serious infections, thromboembolic events, graft rejection, or kidney or neurological toxicity. The longest-treated patient has been insulin-free for over 14 months. The trial has since been extended to 12 subjects, and a second study has been funded including patients with T1D and chronic kidney disease — a population previously excluded from islet transplantation entirely due to tacrolimus nephrotoxicity.
islet transplantation tegoprubart CD40L / CD154 brittle T1D calcineurin inhibitor-free Treg induction insulin independence hypoglycemia unawareness
02 · Significance
Why This Matters to T1D Scientists & Clinicians
For scientists: The CD40–CD40L costimulatory pathway has been a target of interest in transplantation immunology for decades — but earlier anti-CD40L antibodies were abandoned after life-threatening thromboembolic complications. Tegoprubart is a second-generation molecule redesigned to eliminate that risk. This trial is therefore not just a clinical update — it is proof-of-concept that CD40L blockade is viable in humans, that it produces superior islet engraftment by reducing direct beta cell toxicity, and that it generates a more tolerogenic immune environment via Treg expansion. The 3–5x improvement in engraftment efficiency is a striking mechanistic finding with major implications for how we think about the immune-metabolic interface in islet survival.
For clinicians: The implications here are immediate and practice-changing. Every clinician managing patients with brittle T1D knows the impossible calculus: islet transplantation could transform their patients’ lives, but the toxicity of the required immunosuppression makes many patients ineligible or unwilling to proceed. A regimen built around tegoprubart instead of tacrolimus potentially expands the eligible patient population — including, for the first time, patients with pre-existing kidney disease — and removes the neurological, cardiovascular, and metabolic side effects that make current immunosuppression so burdensome. The data showing zero severe hypoglycemic events across six patients post-transplant is clinically extraordinary.
The broader picture: This trial has implications far beyond cadaveric islet transplantation. As stem cell-derived islets (Vertex’s VX-880, Sana Biotechnology, CRISPR Therapeutics) move through clinical trials, the immunosuppression question becomes even more central — you cannot scale beta cell replacement therapy to millions of patients while relying on tacrolimus. Tegoprubart, or the pathway it targets, may be the immunological foundation on which the next generation of T1D cell therapies is built.
03 · Live Discussion
Four Questions We Will Ask the Speaker
Q1
Islet engraftment was estimated to be 3–5x higher with tegoprubart versus tacrolimus. How much of that improvement do you attribute to reduced direct beta cell toxicity versus a more favorable immune environment — and do you have the biomarker data to distinguish between these two mechanisms?
Q2
Earlier anti-CD40L antibodies were halted due to thromboembolic complications. What structural or pharmacological changes in tegoprubart address that risk, and what safety monitoring is in place in this trial to give you and regulators confidence that this concern has been genuinely resolved?
Q3
T1D is an autoimmune disease — these patients’ immune systems attacked their original beta cells. Does tegoprubart’s mechanism address the autoimmune recurrence problem, or does it only protect against allograft rejection? What do you see in your patients’ autoimmune biomarkers over time?
Q4
Looking ahead — do you see tegoprubart as the right immunosuppression backbone for stem cell-derived islet trials, and what would need to be true for it to eventually be used without any other immunosuppressive agents at all?
04 · Go Deeper
4 Key Associated Papers
Shapiro AMJ et al. · 2006
International trial of the Edmonton Protocol for islet transplantation
New England Journal of Medicine, 355:1318–1330
Kenyon NS, Ricordi C et al. · 1999
Long-term survival and function of intrahepatic islet allografts in baboons using anti-CD154 (CD40 ligand) monoclonal antibody
Diabetes, 48(7):1473–1481 — the foundational preclinical anti-CD40L islet study
Vantyghem MC, de Koning EJP et al. · 2019
Advances in beta-cell replacement therapy for the treatment of type 1 diabetes
The Lancet, 394(10205):1274–1285
Eledon Pharmaceuticals · 2024
Tegoprubart mechanism of action: CD40L pathway blockade, costimulation inhibition, and Treg polarization
Eledon Pharmaceuticals Science Overview
05 · TheSugarScience YouTube
4 Videos to Watch Before the Talk
▶ SCI Town Hall: Working With the FDA to Allow Greater Access to Islet Transplant in the US
The regulatory landscape for islet transplantation — essential context for understanding where Witkowski’s trial sits in the broader clinical pathway.
▶ Ask the Expert: Braulio Marfil-Garza, PhD, with James Shapiro, MD — University of Alberta
James Shapiro created the Edmonton Protocol — the foundation on which all modern islet transplantation is built. Indispensable background for this talk.
▶ Ask the Expert: Chris Fraker, PhD & Allison Bayer, PhD — Diabetes Research Institute Foundation
The DRI at University of Miami is Witkowski’s collaborating institution on this trial. Fraker and Bayer cover islet isolation and transplant biology directly relevant to this work.
▶ State of the Science: Clinical Pancreatic Transplant Is Still Relevant for the Future of T1D
A big-picture look at where clinical islet and pancreas transplantation fits in the T1D cure landscape — and why improving immunosuppression is the linchpin.
TheSugarScience · Expediting a cure for T1D by curating the scientific conversation
thesugarscience.org