Biosensor Cell Array Reveals Temporal GABA Secretion Dynamics from Pancreatic Islets- Austin Stis- Phelps Lab- UFlorida
Ahead of the talk May 14
TheSugarScience T1D Th1nk Tank
📅 Registration
Date: Today, May 14, 2026 · 9:00 AM Pacific · 12:00 PM Eastern Format: Free virtual seminar for the global T1D research community — all are welcome.
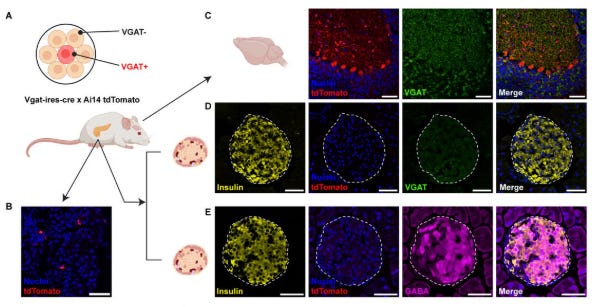
Adapted from Figure 3A from the paper cited.
👤 About the Speaker
Austin Stis is a fourth-year doctoral candidate in the laboratory of Edward Phelps, PhD in the J. Crayton Pruitt Family Department of Biomedical Engineering at the University of Florida. He completed his BSE and MSE in Biomedical Engineering at the University of Michigan, where he trained with Dr. Brendon Baker. He is a fellow of the T32 Interdisciplinary Graduate Program in Type 1 Diabetes & Biomedical Engineering — a program that trains engineers and biologists to work at the intersection of T1D science and technology.
His doctoral research focuses on a molecule that most people associate with the brain, not the pancreas: gamma-aminobutyric acid (GABA). Beta cells synthesize and store GABA at concentrations rivaling those found in inhibitory neurons — yet the mechanism by which they release it, the timing of that release, and its functional role in the islet have remained surprisingly contested. Stis brings an engineer’s toolkit to a biologist’s question — combining specialized neurotransmitter HPLC, genetically encoded biosensing proteins, GABA biosensor cell arrays, and live confocal calcium imaging to resolve GABA secretion dynamics with a temporal precision the field has never had before. He also works with live pancreatic tissue slices from human donors and the CaMPARI2 integrative calcium biosensor to link GABA function to heterogenic islet behavior in donors with and without diabetes.
01 · The Paper
“Biosensor Cell Array Reveals Temporal GABA Secretion Dynamics from Pancreatic Islets” Stis AE, Lazimi CS, Ferreira S & Phelps EA · bioRxiv, April 2026 DOI: 10.64898/2026.03.31.715660
GABA is the brain’s primary inhibitory neurotransmitter. It is also, unexpectedly, one of the most abundant signaling molecules in the pancreatic islet — synthesized by beta cells at concentrations as high as those found in the brain, and implicated in a wide range of paracrine and autocrine functions: modulating alpha cell glucagon secretion, protecting beta cells from apoptosis, and potentially regulating immune cell infiltration in T1D. Understanding how and when GABA is released from beta cells is therefore not a niche biochemical question — it is central to understanding how the islet coordinates its secretory behavior and why that coordination breaks down in diabetes.
The problem is that GABA secretion from islets has been technically difficult to measure with the resolution needed to resolve its dynamics. Bulk biochemical assays and HPLC can measure cumulative GABA output but lose all temporal information. The field has long debated whether GABA is released continuously (tonically) from the cytosol via volume-regulated anion channels, or in regulated pulses co-secreted with insulin from dense-core vesicles. The debate has persisted partly because the tools to settle it didn’t exist.
The Phelps lab built them. The centerpiece of this paper is a GABA biosensor cell array — a specialized detection platform where cells engineered to express a highly sensitive GABA-responsive reporter are arrayed around individual islets, enabling real-time spatial and temporal detection of GABA release with single-islet resolution. Combined with iGABASnFR (a genetically encoded fluorescent GABA sensor) and simultaneous confocal calcium imaging in both the islet and the biosensor cells, the system captures the full spatiotemporal profile of GABA secretion as it happens.
The results resolve the debate — and reveal complexity that neither camp fully anticipated. The key findings:
GABA release is not co-secreted with insulin. Using insulin secretion modulators, the team demonstrated that cumulative GABA release does not track with glucose concentration or insulin secretion — settling a long-standing controversy and ruling out the granule co-release model as the dominant mechanism.
Beta cells do not express VGAT. VGAT reporter mice confirmed that beta cells lack the vesicular GABA transporter required for regulated vesicular release — definitively ruling out the synaptic vesicle model that had been proposed since early work in the field.
GABA is secreted in pulses via LRRC8A/D VRAC. Rather than tonic cytosolic leak, GABA exits beta cells in discrete pulses through the LRRC8A/D isoform of the volume regulatory anion channel — a regulated, channel-mediated mechanism that had been hypothesized but never directly demonstrated with this temporal precision.
GABA pulses are temporally coordinated with calcium oscillations. Simultaneous calcium imaging revealed that GABA release is tightly coupled to the peaks of beta cell calcium oscillations — the same electrical events that trigger insulin secretion — suggesting GABA functions as a rapid feedback signal that strengthens and reinforces the calcium-driven secretory cycle.
A rapid, glucose-triggered GABA pulse precedes sustained release. Immediately following the transition to high glucose, there is a discrete, acute GABA pulse coinciding with the first-phase calcium response — a previously unresolved secretory event confirmed by four independent methods (biosensor cells, iGABASnFR, HPLC of short static incubations, and perifusion fraction analysis).
Tags: GABA · VRAC · LRRC8A · calcium oscillations · biosensor · islet physiology · beta cell · paracrine signaling · neurotransmitter · iGABASnFR · T1D · pancreatic islet · live tissue slice · nPOD
02 · Why This Matters
For scientists: The resolution of the vesicular vs. channel-mediated GABA secretion debate has immediate consequences for how the field interprets decades of prior data and designs future experiments. If GABA is released in calcium-coupled pulses via VRAC rather than tonically from the cytosol or in granules with insulin, then its paracrine and autocrine effects on alpha cells, delta cells, and immune cells are not continuous background signals — they are discrete, temporally structured events synchronized with the beta cell’s electrical activity. This fundamentally changes how we should think about GABA’s role in coordinating islet secretory dynamics and how we interpret perturbations of the GABA system in T1D. The biosensor cell array platform itself is a methodological contribution — a generalizable tool for detecting any secreted small molecule from intact islets with real-time spatial and temporal resolution.
For clinicians: GAD65 — the enzyme that synthesizes GABA in beta cells — is one of the primary autoantigens in T1D. Anti-GAD65 antibodies are among the most clinically used T1D autoantibody biomarkers and are thought to impair GABA production by neutralizing GAD65 activity. If GABA release is temporally coupled to calcium oscillations and serves as a feedback amplifier of secretory coordination, then autoimmune impairment of GAD65 — and therefore GABA synthesis — could disrupt islet secretory coordination well before overt beta cell loss occurs. This positions the GABA system as a potential early functional biomarker of islet dysfunction and raises the question of whether protecting or augmenting GABA signaling could be a therapeutic target in early-stage T1D.
The broader picture: GABA-based therapeutics have been explored in T1D for more than a decade — oral GABA, artemisinins claimed to convert alpha cells to beta cells via GABA signaling, and GABA receptor agonists have all been proposed as therapeutic strategies. Most of these proposals were built on an incomplete and partially incorrect understanding of how GABA is released from the islet. The mechanistic clarity this paper provides — specifically, that GABA release is pulse-like, calcium-coupled, and channel-mediated — gives the field a firm experimental foundation from which to evaluate and redesign GABA-based therapeutic strategies with much greater precision.
03 · Four Questions We Will Ask the Speaker
Q1. GABA pulses are coupled to calcium oscillation peaks — but calcium oscillations themselves are coordinated across the islet via gap junctions, with hub cells playing a pacemaker role. Is GABA release synchronized across the islet spatially, or does it originate from specific subpopulations of beta cells — and does it differ between hub and follower cells?
Q2. Your data shows a rapid, acute GABA pulse coinciding with the first-phase calcium response at glucose stimulation onset. What is the functional consequence of this acute pulse — does it act on alpha cells to suppress glucagon in the first phase, does it feed back onto beta cells to amplify or sustain the calcium response, or both?
Q3. GAD65 is a major T1D autoantigen and anti-GAD65 antibodies impair GABA synthesis. In your live human pancreas tissue slice experiments from T1D donors, do you see measurable differences in GABA secretion dynamics, amplitude, or coupling to calcium oscillations compared to non-diabetic donors — and does the GABA pulse structure break down early in T1D progression?
Q4. The LRRC8A/D VRAC is the channel mediating GABA release in your model. VRAC channels are volume-sensitive and respond to cell swelling. What triggers VRAC opening in a beta cell during glucose stimulation — is it a calcium-dependent mechanism, a metabolic signal, or actual cell volume changes — and could VRAC be a therapeutic target for modulating GABA release?
04 · Four Key Associated Papers
1. Caicedo A & Phelps EA et al. (2020) Mechanism and effects of pulsatile GABA secretion from cytosolic pools in the human beta cell Nature Metabolism, 2:1110–1126 · PMC → The foundational Caicedo/Phelps paper establishing that GABA is secreted in rhythmic bursts from human beta cells independently of glucose — the direct predecessor to today’s paper and the study that motivated the higher-resolution biosensor approach Stis now applies.
2. Hagan DW, Ferreira SM, Santos GJ & Phelps EA (2022) The role of GABA in islet function Frontiers in Endocrinology, 13:972115 · PMC → The Phelps lab’s comprehensive review of GABA biology in the islet — covering synthesis, secretion mechanisms, receptors, and paracrine/autocrine functions. Essential background for understanding the full biological context of today’s secretion dynamics paper.
3. Lazimi CS, Stis AE, Panzer JK, … Phelps EA (2025) Efficient transduction of pancreas tissue slices with genetically encoded calcium integrators bioRxiv · Read preprint → The companion Phelps lab paper developing the CaMPARI2 calcium integrator platform for live pancreas slice studies — the human tissue tool that Stis uses alongside the biosensor array to study GABA dynamics in donor islets with and without diabetes.
4. Braun M, Ramracheya R, Bengtsson M, … Rorsman P (2010) Gamma-aminobutyric acid (GABA) is an autocrine excitatory transmitter in human pancreatic beta-cells Diabetes, 59(7):1694–1701 · PubMed → The landmark Rorsman lab paper proposing that GABA is co-released with insulin from beta cell granules and acts as an autocrine excitatory signal — the prevailing model that today’s paper challenges and partially overturns with higher-resolution temporal data.
05 · Four Videos to Watch First
▶ Ask the Expert: Ed Phelps, PhD — University of Florida Today’s speaker’s PhD mentor — watch this first to understand the Phelps lab’s islet biosensing platform, GABA research program, and the engineering-driven approach to islet biology that Austin Stis has been trained in.
▶ Ask the Expert: Julia Panzer, PhD, with Alejandro Caicedo, PhD — University of Miami Caicedo is the senior author on the foundational pulsatile GABA secretion paper (Nature Metabolism 2020) and a key collaborator in the field — Panzer and Caicedo provide the live tissue slice and human islet context that Stis’s work directly builds on.
▶ Ask the Expert: Matthew Merrins, PhD & Richard Benninger, PhD — 3D Light-Sheet Imaging of the Islet Merrins and Benninger are leaders in islet calcium imaging and electrical coordination — the same calcium oscillation dynamics that Stis shows are temporally coupled to GABA release. Essential methodological and biological context.
▶ Ask the Expert: Zachary Freyberg, MD PhD — University of Pittsburgh Freyberg studies vesicular neurotransmitter release and secretory vesicle biology in beta cells — the mechanistic alternative to the VRAC channel-mediated GABA release that Stis’s paper establishes, and the prior-art framework his findings now update.
TheSugarScience · Expediting a cure for T1D by curating the scientific conversation · thesugarscience.org