Advancing Suspension Manufacturing of Stem Cell–Derived Islets in Vertical Wheel Bioreactors for Scalable Diabetes Therapy= Nidheesh Dadheech PhD, SUNY Upstate Medical
Pregame: Ahead of the April 2 talk
TheSugarScience T1D Th1nk Tank
📅 Registration
Format: Free virtual seminar for the global T1D research community — all are welcome.
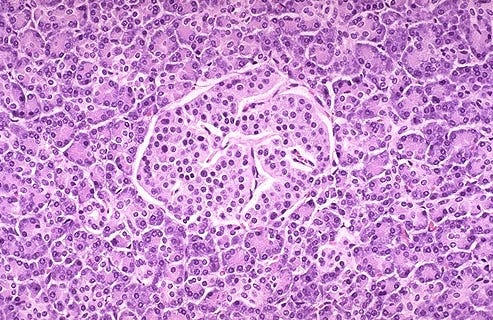
Adapted from Dadheech et al., npj Regenerative Medicine (2025), Fig. 3J. DOI: 10.1038/s41536-025-00409-y
About the Speaker
Nidheesh Dadheech, PhD is an Assistant Professor of Medicine and Director of the Institute of Regenerative Medicine at SUNY Upstate Medical University in New York, where his Dadheech Stem Cell Islet Bioengineering Laboratory focuses on developing human iPSC-derived islet cell products for the treatment of all forms of diabetes — without immunosuppression. His research investigates the human genetics and genomics of patient-derived pluripotent stem cells and differentiated islets, with particular attention to resolving the cellular and genomic heterogeneity that currently limits successful clinical translation.
He earned his PhD in Biochemistry from The Maharaja Sayajirao University of Baroda, India, and completed postdoctoral studies at the Alberta Diabetes Institute, University of Alberta, studying molecular regulation of beta cell mass expansion and regeneration. He subsequently served as Senior Cell Therapy Lead in the laboratory of Dr. A.M. James Shapiro — the pioneering surgeon-scientist behind the Edmonton Protocol — within the Clinical Islet Transplant Program at Alberta, contributing directly to the design of next-generation SC-islet therapies. He is the recipient of the Researcher of the Year Award from the Alberta Diabetes Foundation (Hummingbird Gala 2025), the Young Investigator Award from the New York Stem Cell Foundation, and a Canadian Commonwealth Scholarship from the Government of Canada.
01 · The Paper
“Scale-up Manufacturing Approach for Production of Human Induced Pluripotent Stem Cell–Derived Islets Using Vertical Wheel® Bioreactors” Dadheech N, Bermúdez de León M, Czarnecka Z, Cuesta-Gomez N, Tewari Jasra I, Pawlick R, Marfil-Garza B, … MacDonald P & Shapiro AMJ · npj Regenerative Medicine, 2025 DOI: 10.1038/s41536-025-00409-y
The science of making stem cell-derived beta cells has advanced dramatically. The manufacturing science — how to produce them at the quantities, consistency, and cost required to treat millions of people — has lagged behind. This paper addresses that gap head-on, demonstrating a complete, suspension-based differentiation platform for iPSC-to-islet conversion inside Vertical Wheel® (VW) bioreactors that achieves meaningful clinical-scale yields without sacrificing product quality.
The problem Dadheech’s team set out to solve is deeply practical. Current SC-islet differentiation protocols fall into two categories: planar 2D cultures on flat plates, which are inherently low-throughput and labor-intensive; or hybrid approaches that begin in 2D and shift to 3D suspension only in later stages. Both suffer from cell loss during media changes, batch-to-batch inconsistency driven by the heterogeneous shear forces in conventional stirred-tank bioreactors, and fundamental limits on scalability. The Vertical Wheel bioreactor was designed to address these problems — its unique rotating impeller geometry generates low, uniform shear throughout the vessel, which is critical for the fragile 3D cluster aggregates that form during islet differentiation.
Dadheech’s group developed a complete, all-suspension 27-day, six-stage differentiation protocol (S1: definitive endoderm → S2: primitive gut tube → S3: posterior foregut → S4: pancreatic progenitors → S5: endocrine progenitors → S6: SC-islets) implemented entirely within VW bioreactors from the first day of differentiation. This is a meaningful advance: doing the entire process in suspension eliminates the cell-loss and consistency problems associated with transitioning between culture formats mid-protocol.
The headline result is a 12-fold increase in islet equivalent count (IEQ) when scaling from 0.1 L to 0.5 L vessels — from ~15,000 to ~183,000 IEQs — with only a 5x increase in vessel volume. Critically, this scale-up did not compromise product quality: SC-islets from both vessel sizes showed equivalent morphology, size distribution, and transcriptional signatures. Beta cell composition reached ~63% (C-peptide⁺/NKX6.1⁺/ISL1⁺), glucose-stimulated insulin secretion showed a 3.9–6.1-fold stimulation index, and single-cell RNA sequencing confirmed transcriptional maturity comparable to adult donor islets. The protocol was validated across three distinct iPSC lines — from healthy donors, total pancreatectomy patients, and T1D donors — demonstrating robustness across the genetic diversity that a clinical program would encounter.
The functional proof came in vivo: SC-islets transplanted into STZ-diabetic SCID-beige mice reversed hyperglycemia in more than 50% of animals within 30 days and in all transplanted mice by 60 days. Harvested grafts at 2, 3, and 4 months showed continued in vivo maturation and improved transcriptomic signatures over time — suggesting the VW-manufactured product continues to develop functionally after transplantation, rather than plateauing.
Tags: iPSC-derived islets · bioreactor · scale-up manufacturing · Vertical Wheel · suspension culture · SC-islets · IEQ · clinical-grade manufacturing · diabetes cell therapy · single-cell RNA sequencing · GMP
02 · Why This Matters
For scientists: Manufacturing is not a downstream afterthought in cell therapy — it is a core scientific problem that shapes everything from product consistency to translational feasibility. The VW bioreactor platform addresses three persistent failure modes in SC-islet manufacturing simultaneously: the shear damage that disrupts 3D cluster formation in stirred-tank systems, the cell loss and protocol-switching challenges that occur when moving between 2D and 3D formats mid-differentiation, and the batch heterogeneity that makes regulatory approval difficult. The 12-fold IEQ yield gain at only 5x vessel scale is particularly important — it means each incremental increase in reactor size produces disproportionately more product, which is exactly the economic and logistical behavior needed for clinical manufacturing. The scRNA-seq validation across three iPSC lines also sets a new standard for characterizing manufacturing output.
For clinicians: The numbers matter here. A single adult T1D patient requires approximately 400,000–500,000 IEQs for a therapeutic islet transplant. The Dadheech protocol produces ~183,000 IEQs from a 0.5 L vessel. The pathway to therapeutic dose — and beyond, to multi-patient manufacturing from a single batch — is now clearly visible and mathematically tractable. The fact that the protocol works across iPSC lines from T1D donors is especially significant: it means patient-specific, autologous SC-islet products — potentially manufactured without the need for immunosuppression — are within reach of a practical production workflow. For programs like Shapiro’s at Alberta, where clinical islet transplantation is already standard care, this bioreactor platform provides the missing manufacturing link between laboratory-scale differentiation science and clinical-grade cell therapy.
The broader picture: The SC-islet field is at an inflection point. Vertex’s Zimislecel and Sana’s hypoimmune islet programs have demonstrated proof of concept in humans. The next competitive frontier is not whether SC-islets can work, but whether they can be made efficiently, consistently, and affordably enough to reach more than a handful of clinical trial participants. Manufacturing platforms like the VW bioreactor system are the infrastructure the field needs — and this paper provides the most detailed, openly published blueprint yet for how to build it.
03 · Four Questions We Will Ask the Speaker
Q1. The 12-fold IEQ increase going from 0.1 L to 0.5 L is impressive — but clinical programs will need to reach 2 L, 10 L, and beyond. Have you begun testing at larger vessel sizes, and does the IEQ yield continue to scale superlinearly, or does it plateau at a certain reactor volume?
Q2. You validated the protocol across three iPSC lines — healthy, pancreatectomy, and T1D donors. iPSC lines from different donors are known to vary significantly in their differentiation efficiency and epigenetic memory. What is the variability in IEQ yield and beta cell purity across additional donor lines, and what quality control release criteria would you propose for a clinical-grade batch?
Q3. The scRNA-seq data shows transcriptional maturity comparable to adult islets, but transplanted SC-islets continue to mature in vivo over months. What are the transcriptional differences that remain between your VW-manufactured SC-islets and adult donor islets, and what modifications to the protocol — extended culture, circadian entrainment, or additional maturation factors — might close that gap before transplantation?
Q4. Your background includes work on immunosuppression-free strategies from the Shapiro Lab. How do you envision combining the VW manufacturing platform with genetic engineering approaches — such as Rnls deletion or hypoimmune modifications — and does the all-suspension format impose any constraints on incorporating gene editing steps into the protocol?
04 · Four Key Associated Papers
1. Verhoeff K, Cuesta-Gomez N, Dadheech N, … Shapiro AMJ (2025) Scalable bioreactor-based suspension approach to generate stem cell–derived islets from healthy donor-derived iPSCs Transplantation, 109(1):e22–e35 · Read paper → The companion paper from the same Alberta team — the earlier characterization study that established the foundational VW bioreactor suspension differentiation approach and set the stage for the scale-up advances in today’s npj paper.
2. Shapiro AMJ, Lakey JRT, Ryan EA et al. (2000) Islet transplantation in seven patients with type 1 diabetes mellitus using a glucocorticoid-free immunosuppressive regimen New England Journal of Medicine, 343:230–238 · PubMed → The Edmonton Protocol — the landmark paper by Dadheech’s mentor James Shapiro that proved islet transplantation could reverse T1D, and established the clinical benchmark that SC-islet manufacturing must ultimately match.
3. Balboa D, Barsby T, Lithovius V, … Otonkoski T et al. (2022) Functional, metabolic and transcriptional maturation of human pancreatic islets derived from stem cells Nature Biotechnology, 40:1042–1055 · PubMed →** The leading reference on the transcriptional and functional maturation benchmarks for SC-islets — the quality standard against which the VW-manufactured product in today’s paper is compared.
4. Millman JR, Xie C, Van Dervort A, … Melton DA (2016) Generation of stem cell–derived β-cells from patients with type 1 diabetes Nature Communications, 7:11463 · PubMed → Proof that patient-specific iPSC lines from T1D donors can be differentiated into functional SC-islets — the scientific rationale for Dadheech’s inclusion of T1D donor iPSC lines in his manufacturing validation.
05 · Four Videos to Watch First
▶ State of the Science: Fast Tracking Stem Cell–Derived Islets to the Clinic — What Is Needed? The essential big-picture primer on where the SC-islet field stands and what barriers — including manufacturing scale — remain. Watch this first.
▶ Ask the Expert: Braulio Marfil-Garza, PhD, with James Shapiro, MD — University of Alberta Dadheech’s direct collaborators from the Shapiro Lab at Alberta — the clinical islet transplant program where this manufacturing work was developed. Essential background on the clinical context this platform is designed to serve.
▶ Ask the Expert: Jeffrey Millman, PhD — Washington University in St. Louis Millman is one of the field’s leaders on SC-islet differentiation protocols — his work defines the quality benchmarks that today’s VW bioreactor platform is designed to meet at clinical scale.
▶ T1D Th1nk Tank: Peter Thompson, PhD, & Jasmine Pipella, PhD — University of Manitoba Thompson and Pipella work on the transplant immunology and clinical translation of islet therapies — the downstream clinical program that manufacturing advances like Dadheech’s directly enable.
TheSugarScience · Expediting a cure for T1D by curating the scientific conversation · thesugarscience.org